Main page (Russian)
 Search in database (English)
Search in database (English)
Properties of substance:
ninhydrin
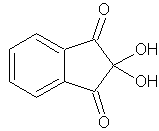
skc-fileSynonyms:
2,2-dihydroxy-1,3-indanedione
Group of substances:
organic
Physical appearance:
prismatic crystalsEmpirical formula (Hill's system for organic substances):
C9H6O4Molar/atomic mass: 178.14
Boiling point (°C):
242Solubility (g/100 g of solvent):
diethyl ether: sparingly soluble [Ref.]
ethanol: sparingly soluble [Ref.]
water: very soluble [Ref.]
Synthesis 1:
Reference: Becker, H.-D., & Russell, G. A. Synthesis of Ninhydrin / Journal of Organic Chemistry. - 1963. - Vol. 28, No. 7 pp. 1896 [doi: 10.1021/jo01042a502]
One gram of 2-chloro-2-methylmercapto-1,3-indandione was added slowly to 50 ml. of boiling water in a 100-ml. erlenmeyer flask. The slightly yellow solution was kept on a steam bath for 12 hr. during which most of the water evaporated. The concentrated aqueous solution was transferred to a 50-ml. beaker and evaporated on a steam bath for another hour to yield a crystalline residue which was dried under vacuum. The material thus prepared (775 mg., 99%) had m.p. 239—240° and an infrared spectrum identical with that of of commercial ninhydrin.Synthesis 2:
Reference: Teeters, W. O., & Shriner, R. L. A New Preparation of Ninhydrin / Journal of the American Chemical Society. - 1933. - Vol. 55, No. 7 pp. 3027-3028 [doi: 10.1021/ja01334a072]
In a 2-liter three-necked flask fitted with a reflux condenser and mechanical stirrer was placed 55 g. of sublimed selenium dioxide dissolved in 1200 cc. of dioxane and 25 cc. of water. The stirrer was started and the solution heated to approximately 60—70°. The flame was withdrawn, 73 g. of crude 1,3-indanedione was added and the resulting mixture refluxed for six hours. A solid separated during this period and was filtered off while the mixture was still hot. The filtrate was transferred to a distilling flask and three-fourths of the dioxane distilled. Between 400 and 500 cc. of water was added and the solution boiled to coagulate the tarry precipitate, which was then removed by filtration. The filtrate was concentrated by distillation to approximately 250 cc. and filtered. The filtrate was boiled with 1 g. of norite, filtered again, concentrated to 125 cc. and allowed to stand at room temperature. The crude ninhydrin which crystallized was filtered, the mother liquor concentrated and a second crop of crystals obtained; total yield of crude, 36—38 g. The impure ninhydrin was contaminated with a trace of selenious acid which acted as a bleaching agent and prevented the formation of the characteristic blue color reaction with α-amino acids. It was purified by recrystallization from hot water with the aid of norite. Long colorless prisms of ninhydrin were obtained by crystallization at room temperature. The yield of the pure ninhydrin was 28—31 g. (31—35% of the theoretical). The product gave none of the customary tests for selenium and gave the characteristic color reactions with α-amino acids. It lost water of hydration and turned red between 125—130° and finally melted with decomposition at 241—243°.
Anal. Calcd. for C9H4O3·H2O: C, 60.67; H, 3.37. Found: C, 60.83; H, 3.64.
The condensation product with o-phenylenediamine was prepared as a derivative and found to melt at 219—220°, which checked the value reported by Ruhemann.
Synthesis 3:
Reference: Heffner, R. J., & Joullie, M. M. Synthetic Routes to Ninhydrins. Preparation of Ninhydrin, 5-Methoxyninhydrin, and 5-(Methylthio)Ninhydrin / Synthetic Communications. - 1991. - Vol. 21, No. 21 pp. 2254 [doi: 10.1080/00397919108055457]To a solution of 3-bromo-1-indenone (0.50 g, 2.8 mmol) in benzene (10 mL) was added dimethylsulfoxide (10 mL), followed by the addition of bromine (0.23 g, 1.4 mmol). The solution was heated at reflux, and after six hours was cooled to 25 °C. The reaction was then diluted with water (20 mL). The aqueous phase was then extracted with ethyl acetate (4 x 10 mL) and organic extracts were combined, washed with water (5 mL), dried over Na2SO4, filtered, and concentrated under reduced pressure to yield a solid. This material was recrystallized from water to provide ninhydrin (0.25 g, 60% yield): mp 240 °C, lit. mp 241 °C.
Rf 0.61 methylene chloride : methanol (90 : 10).
IR (KBr): 3530-2900 (broad), 1745 (strong), 1750 (strong), 1715 (strong), 1590 (medium), 1385 (medium), 1290 (weak), 1255 (weak), 1185 (strong), 1150 (medium), 1080 (medium), 1060 (medium), 1010 (medium), 1000 (medium), 940 (strong), 880 (weak), 740 (strong) cm-1.
1H NMR (DMSO-d6): δ 7.58 (2H, s), 8.04-8.10 (4H, m); 1H NMR (DMSO-d6, D2O): 8.04-8.12 (4H, m).
Dissociation:
pKa (1) = 8.6 (25°C, water)
References:
- CRC Handbook of Chemistry and Physics. - 95ed. - CRC Press, 2014. - pp. 3-320
- Dictionary of organic compounds. - Vol. 3, naphthacarbazole - zygadenine. - М.: ИИЛ, 1949. - pp. 947
- Рабинович В.А., Хавин З.Я. Краткий химический справочник. - Л.: Химия, 1977. - pp. 165 [Russian]
- Химический энциклопедический словарь. - Под ред. Кнунянц И.Л. - М.: Советская энциклопедия, 1983. - pp. 379 [Russian]
Your requests if no data into database
© Collected Ruslan Anatolievich Kiper, burewestnik@mail.ru



